Lay-Language Papers
C-Arm Cone-Beam CT for Image-Guided Interventions
Jeff Siewerdsen (jsiewerd@uhnres.utoronto.ca)
M. Daly, D. Moseley, Y. Cho, S. Ansell, G. Wilson, D. Jaffray
University Health Network, Siemens Medical Solutions
Lay language description of papers WE-B-L100F-2 / TH-C-M100J-6 / TH-D-L100J-8
Wednesday, July 25 and Thursday, July 26
2007 AAPM Meeting, Minneapolis, MN
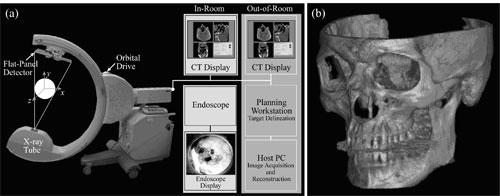
Figure 1. (a) Illustration of the mobile isocentric C-arm modified to include a flat-panel detector, motorized orbit, geometric calibration, and control system for CBCT acquisition. Intra-operative CBCT reconstructions provided by the host PC are displayed on an in-room display. (b) The example image (skull) illustrates the 3D spatial resolution and FOV.
Image-guided interventions have conventionally relied on image data acquired before the procedure from a diagnostic computed tomography (CT) or magnetic resonance (MR) scanner. Within the operating theater, the surgeon or interventional radiologist would then utilize a real-time navigation system to navigate within the preoperative image data. This approach has offered a tremendous advance in image-guided interventions over the last 20 years, with greatest impact in orthopedic surgery and neurosurgery. But reliance on preoperative images limits the application of image-guided interventions to procedures that have surgical targets and surrounding critical anatomies that do not significantly deform during the procedure. Such an approach also does not allow visualization of changes imparted during the procedure, such as visualizing residual tumor after excision.
Intraoperative imaging promises to overcome these conventional limitations by providing the surgeon with image updates during the procedure. Technologies such as interventional MR, CT and others are under development to accurately depict patient anatomy during the procedure by providing second-by-second 3-D image data. A new technology showing particular promise is cone-beam CT (CBCT) implemented on a surgical C-arm.
Research at the University of Toronto, headed by Dr. Jeffrey Siewerdsen, in collaboration with clinical researchers at the University Health Network (UHN) and at Siemens Medical Solutions (SMS), has yielded a 3-D intraoperative imaging technology that could provide surgeons with sub-millimeter spatial resolution and soft tissue visibility in near real-time. The technology involves the development of cone-beam CT on a mobile C-arm to acquire a full volume image in a single rotation about the patient.
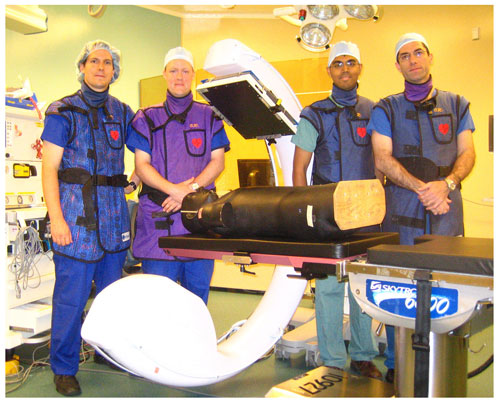
Figure 2. Photograph of the CBCT-capable C-arm in the operating room. Dr. Siewerdsen (left) has led the research effort at UHN over the last 5 years, with engineers (Michael Daly and Sajendra Nithiananthan) working to optimize image quality, minimize radiation dose, and integrate with systems for real-time surgical navigation. Clinical collaborators such as Dr. Gideon Bachar, (right) are deploying the system in clinical research trials for the first time in precise image-guided procedures guided by CBCT.
Proof of principle for this technology began in 2000. Physicists and engineers worked together to modify a commercially available C-arm, the Siemens PowerMobil, for CBCT capability. Collaboration between physicists and clinicians at UHN and engineers at SMS explored early clinical applications of this technology ranging from orthopedic surgery to brachytherapy. Today, the CBCT-capable C-arm is under investigation for a broad spectrum of image-guided interventions, ranging from head-and-neck surgery to spine, thoracic, breast, abdominal, and prostate surgery as well as interventional radiology. Because the C-arm is mobile, it also provides a valuable 3-D imaging technology well suited for emergency rooms and intensive care units.
The key physical imaging performance characteristics of C-arm CBCT are the capability for sub-millimeter spatial resolution and the ability to view soft tissue, each obtained at an imaging dose which is a small fraction of that from a diagnostic CT scan. While the image quality achieved with early the C-arm prototype is not quite equivalent to that of a diagnostic CT scanner, image quality has been shown to be sufficient to guide surgeons and radiologists with respect to soft-tissue targets and critical structures.
The CBCT C-arm is made possible by the development of three distinct technologies: CBCT reconstruction, flat-panel detectors, and the isocentric C-arm. CBCT reconstruction techniques are similar to conventional CT reconstruction techniques, except that an entire volume, rather than just a few slices, is reconstructed from a single orbit about the patient. Flat-panel detectors developed in the 1990s provide digital x-ray imaging technology with the efficiency, speed and image quality well-suited for CBCT. The C-arm is isocentric, meaning that it maintains a fixed field of view as the C-arm gantry moves about the patient, and therefore provides an important platform for acquiring images for CBCT. Together, these technologies provide an important new addition to the arsenal of image-guided surgery, providing multi-mode fluoroscopy and 3-D imaging in the operating room.
The early proof-of-principle research was conducted by UHN physicists Dr. Jeffrey Siewerdsen and Dr. David Jaffray. They collaborated with SMS scientists and engineers, including Drs. Klaus Hermann, Rainer Graumann, Dieter Ritter, and Matthias Mitschke. Further development and the first clinical application of the C-arm took place over the last five years at UHN under the direction of Dr. Siewerdsen in collaboration with Dr. Jonathan Irish, chief of surgical oncology, Dr. Walter Kucharczyk, head of neuroradiology, and Dr. Brian Wilson, head of biophysics and bioimaging. A team of engineers, including Michael Daly, Steve Ansell, Graham Wilson, and Sajendra Nithiananthan, helped to develop streamlined, high-performance techniques for C-arm CBCT, and the system is beginning clinical trials at UHN.
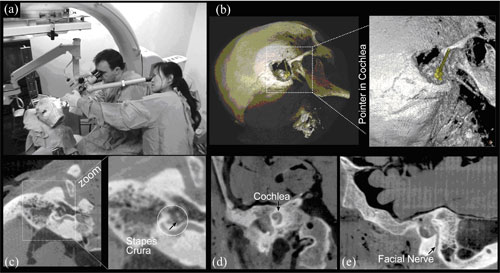
Figure 3. C-arm CBCT illustrated in the context of temporal bone surgery. (a) Experimental setup for early cadaver studies. Illustration of CBCT image quality in (b) volume renderings of the skull following mastoidectomy, with a pointer placed in the cochlea, (c) the stapes crura (among the finest bones in the human body), (d) the cochlea, and (e) the facial nerve.
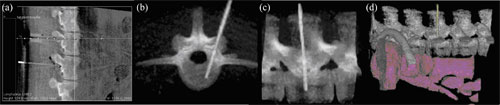
Figure 4. C-arm CBCT for image-guided spinal procedures. Images show CBCT renderings of an anesthetized pig (~40 kg) undergoing transpedicular placement of fiber optic probes for photodynamic therapy of spinal metastases.
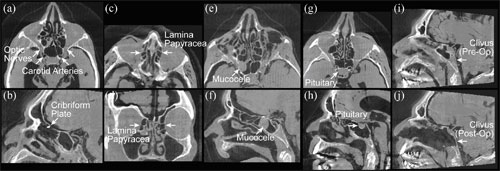
Figure 5. Anatomical landmarks in head and neck surgery identified in CBCT images. (a-d) Example critical structures. (e-h) Example target structures. (i-j) Pre- and post-operative views of the clivus. Note the exquisite posterior margin following CBCT-guided ablation.
The future for such technology entering more routine clinical practice is immediate, and the value of high-quality 3D intraoperative imaging is clear, but not without remaining challenges. Such technology supports broader innovation in the development of advanced image analysis techniques (e.g., 3D deformable image registration), advanced treatment delivery systems (e.g., robot-assisted surgery and novel ablative therapies), as well as the integration of interventional modalities, such as surgery and radiation therapy, with 3D imaging providing a common ground for integrated treatment planning, guidance, and response assessment. Intraoperative CBCT will almost certainly improve surgical performance and expand the application of minimally invasive interventions to cases that would be otherwise untreatable.
Associated AAPM Meeting Presentations: Wednesday, July 25, 8:55 AM (WE-B-L100F-2), Thursday, July 26, 11 AM (TH-C-M100J-6 ), and 2:54 PM (TH-D-L100J-8)