An AAPM Grand Challenge
Overview
AAPM is sponsoring a Challenge on the calculation of time-integrated activity in radiopharmaceutical therapy dosimetry. The results of the Challenge will be announced publicly at the Grand Challenge Symposium of the 2023 AAPM Annual Meeting in Houston, TX.
Background
 Dosimetry for radiopharmaceutical therapy (RPT) is a complex task. In contrast to external beam radiotherapy, where radiation is deposited through a well-defined external radiation source, the dose distribution in RPT depends on the time-varying distribution of the radiopharmaceutical inside the patient after administration, which is highly dependent on the radiopharmaceutical’s kinetics.
Dosimetry for radiopharmaceutical therapy (RPT) is a complex task. In contrast to external beam radiotherapy, where radiation is deposited through a well-defined external radiation source, the dose distribution in RPT depends on the time-varying distribution of the radiopharmaceutical inside the patient after administration, which is highly dependent on the radiopharmaceutical’s kinetics.
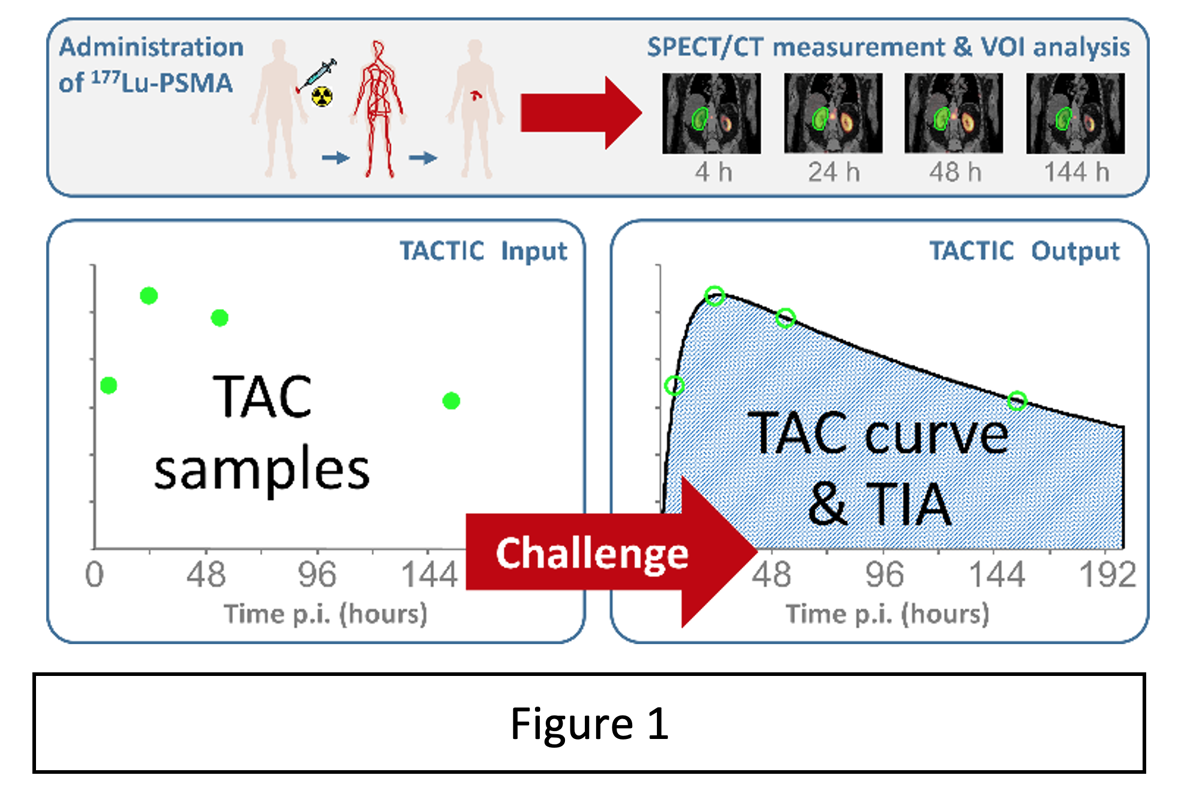
In addition to the geometry and composition of relevant source and target regions and S values describing the physical process of radiation deposition, it is therefore essential for RPT dosimetry to monitor the time course of the radiopharmaceutical distribution in each relevant region. In practice, this translates to a need for accurate activity measurements at different time points post-injection (e.g., by blood samples and/or SPECT/CT imaging) in combination with a fast and repeatable methodology for source or target region delineation, which is illustrated in Figure 1. This results in time-activity curves (TACs) for all relevant structures, based on which time-integrated activities (TIAs) can be calculated.
Goal of the Challenge
Results from the SNMMI 177Lu Dosimetry Challenge 2021 [Uribe et al., JNM 2021] have recently demonstrated that RPT dosimetry is overwhelmingly user- and software-dependent: there is a wide variation in the RPT dosimetry methodology, and understanding the contributions of individual steps within the workflow is crucial for standardization. The TACTIC challenge zooms in on one of the most important sub-aspects of RPT dosimetry: TAC determination. Participants will be presented with a series of snapshots of precisely known time-activity curves at clinically relevant sampling time points. Based on their submitted TAC functions and TIA values, the Challenge Team will assess the degree of variation between different means of TAC/TIA estimation. Among all submissions, we will identify the best-performing methods including their uncertainties and thus take a decisive step towards improved robustness of TAC/TIA determination for RPTs.
Important Dates
- Challenge platform opens for pre-registration: January 19, 2023
- Phase 0 begins, the associated data is available for download: February 15, 2023
- Deadline for submission of Phase 0 results: March 15, 2023
- Phase 1 starts. Partial results of Phase 0 are published online (Challenge webpage): March 16, 2023
- Deadline for submission of Phase 1 results: April 13, 2023
- Phase 2 starts. Partial results of Phase 1 are published online (Challenge webpage): April 14, 2023
- Deadline for submission of Phase 2 results: May 15, 2023
- Winning teams from Phase 1 and 2 are notified of Challenge results: June 1, 2023
- Winning results presented at the Grand Challenges Symposium session of the 2023 AAPM Annual Meeting in Houston, TX (July 23-27, 2023).
Dataset
The Challenge dataset will be composed of 25 synthetic datasets of peri-therapeutic time-activity curves in kidneys, blood, and tumor after 177Lu-PSMA therapy. These data are generated based on randomly selected high-quality clinical datasets, with realistic uncertainties added.
Challenge Format
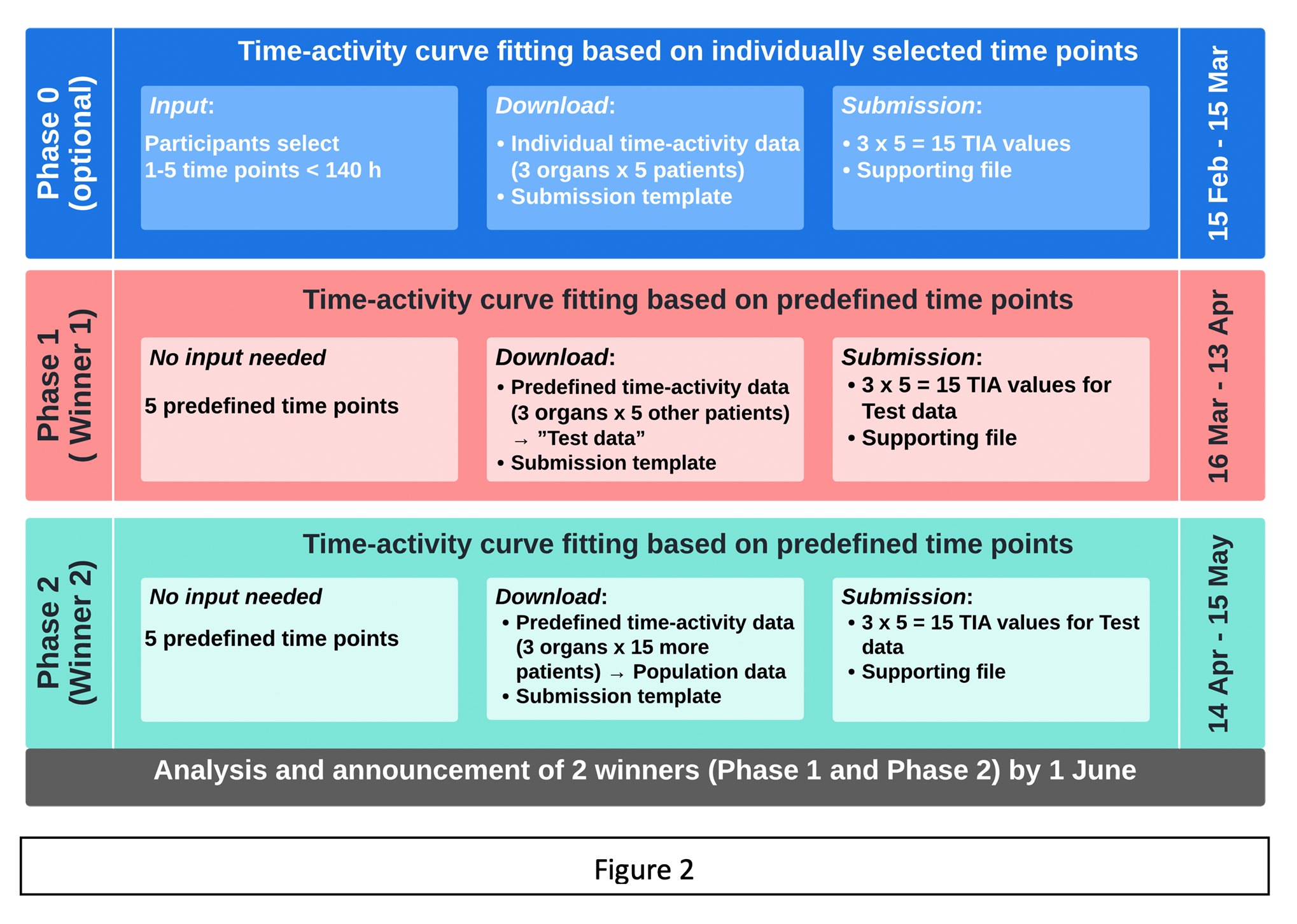
The TACTIC Challenge consists of 3 phases (see Figure 2):
- Phase 0 (15th February – 15th March) [optional warm-up exercise]
Participants may select between 1 and 5 time points from a synthetic predefined dataset of 140 time points < 140 hours post injection of 177Lu-PSMA. A download will be initiated, providing the participants with percent (%) activity data for 3 organs (blood, tumor and kidneys) from 5 separate patients for the time points they selected. A maximum of 3 (organs) x 5 (patients) x 5 (time points) = 75 data points will be available for download. A submission template will also be provided on download.
Participants will then:- Fit the TAC function that best represents the given data points
- Calculate the TIA values based on each fitted TAC function
- Complete the submission template file
- Submit the results back to the Challenge webpage
- Phase 1 (16th March – 13th April)
All participants will download the same predefined set of time-activity data corresponding to a clinical data acquisition scheme, consisting of 5 time points for 3 organs (blood, tumor and kidneys) for 5 patients (other than those used in Phase 0). A submission template will also be provided.
Participants will then:- Fit the TAC function that best represents the given data points
- Calculate the TIA values based on each fitted TAC function
- Complete the submission template file
- Submit the results back to the Challenge webpage
- Phase 2 (14th April – 15th May)
All participants will download the same predefined set of time-activity data corresponding to a clinical data acquisition scheme, consisting of 5 time points for 3 organs (blood, tumor and kidneys) for 15 additional patients (other than those used in Phase 1). A submission template will also be provided. In addition, supporting literature is provided describing state-of-the-art population-based time-activity data fitting.
Participants will then:- Fit the TAC functions that best represent the given data points
- Calculate the TIA values based on each fitted TAC function
- Complete the submission template file
- Submit the results back to the Challenge webpage
- Analysis
All valid submissions from Phases 1 and 2 will be scored by the Challenge Team, who will compare results to the ground truth. The participants with the best score in Phase 1 and Phase 2 will be announced as the two winners. These winners will be awarded complimentary registration to attend the AAPM Annual Meeting 2023 in Houston, TX, where they will present on their methodology in a dedicated Grand Challenge Symposium (in-person attendance mandatory).
Publication Plan
The results of this Grand Challenge will be summarized to reflect the variability and information-content dependence of TAC and TIA values in RPT dosimetry, including suggestions on how to harmonize their calculation. These findings will be published, with participants listed as co-authors, in suitable journals such as Medical Physics or the Journal of Nuclear Medicine, within 2 years from completion of the challenge.
Organizers
- Dr. Marta Cremonesi, European Institute of Oncology, Italy
- Prof. Dr. Gerhard Glatting, Ulm University Hospital, Germany
- Dr. Elisa Grassi, AUSL-IRCCS of Reggio Emilia, Italy
- Dr. Oleksandra V. Ivashchenko, University Medical Center Groningen, the Netherlands
- Dr. Eero Hippelainen, University of Helsinki and Helsinki University Hospital, Finland
- Dr. Deni Hardiansyah, Universitas Indonesia, Indonesia
- Dr. Jan W. T. Heemskerk, Leiden University Medical Center, the Netherlands
- Dr. Jim O’Doherty, Siemens Medical Solutions/Medical University of South Carolina, USA
- Dr. Johannes Tran-Gia, University Hospital Würzburg, Germany
- the AAPM Working Group on Grand Challenges
Contacts
For further information, please contact Sasha Ivashchenko.